Helium bohr model8/15/2023 
Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics. The outermost shell in the Bohr diagram of Beryllium contains 2 electrons that also called valence electrons. This nucleus is surrounded by two-electron shells named K-shell and L-shell. When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). The Bohr Model of Beryllium (Be) has a nucleus that contains 5 neutrons and 4 protons. Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na . As a result of losing a negatively-charged electron, they become positively-charged ions. This figure shows one quater of the orbitals. This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. Two same-shaped orbital planes of electrons are perpendicular to each other. In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). 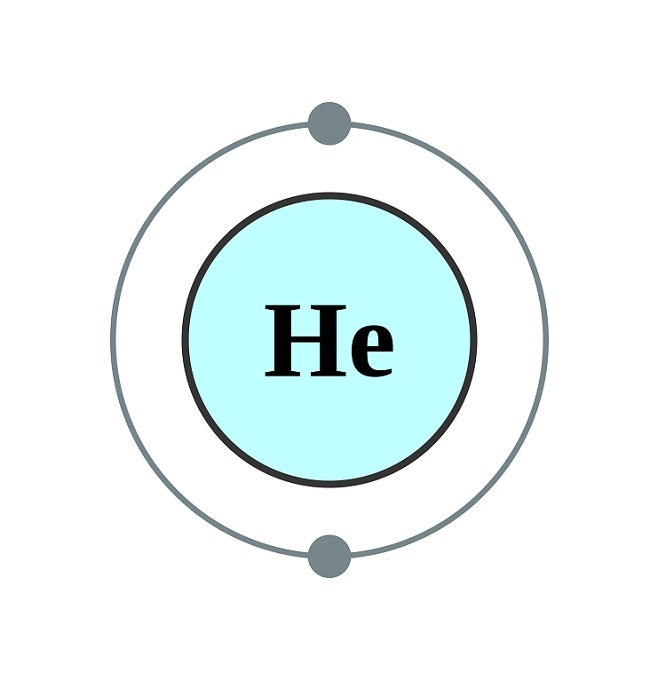
As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. The magnetic moment is zero.Īpplication of the Quantum Theory to Coupled Electrons.-The success of the semi-circular model in giving the correct value for the ionizing potential of helium suggests that in the case of coupled electrons the quantum theory should be applied not to the momentum of the individual electrons according to the relation ∫ pdq = h 2 π, but rather to the momentum which, by being relayed from one electron to another, passes in each direction around the nucleus.\):īohr diagrams indicate how many electrons fill each principal shell. The total energy comes out less than for the Bohr model, and the ionizing potential, 25.62 volts, agrees closely with the experimental value. Assuming the maximum angular momentum of each equal to h 2 π, the absolute dimensions of the model were computed. (3) In the semi-circular model each electron is assumed to oscillate back and forth along an approximately semi-circular path in accord with classical mechanics, each being brought to rest at the end of its path by the repulsion of the other. Another objection is that the magnetic moment is not zero. The model, however, is unstable, for the ionizing potential, computed by applying the quantum theory, comes out negative. (2) In the double circle model the two electrons are assumed to move in two separate, parallel, circular orbits. Accordingly two new models are considered. Bohr became convinced of its validity and spent part of 1912 at Rutherford’s laboratory. Now the chemical evidence suggests that each electron in an atom has its own separate orbit, and that these orbits are closely interrelated. The great Danish physicist Niels Bohr (18851962) made immediate use of Rutherford’s planetary model of the atom. Models for the Helium Atom.-(1) Bohr's model is unsatisfactory because it gives too great a value for the ionizing potential and is not in accord with the optical and magnetic properties of helium.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
